More than ten days after the fire, during the night of 25 to 26 September, at the Lubrizol factory, classified as a Seveso site, the harvesting of fruit and vegetables was banned in around a hundred towns in Normandy and the Hauts-de-France region. Doubts, mistrust, and grey areas remain among the population on the so-called "transparency" elements given by the government. What overall strategy can be put in place to assess the dangers, risks and safety associated with exposure to chemicals? How can the risks be perceived and the expectations of civil society be met? As the Greenfacts analysis shows, nothing is simple. And European regulations are complicated and almost always inaudible or even inappropriate for citizens. Deciphering.
Context: the difficulty of going beyond the limits of our "connai-sciences".
As individuals, we often find it difficult to change our well-established opinions, even when new facts or new knowledge of proven facts should lead us to do so. As some of René Magritte's paintings warn us: " This is not a pipe! "However, we know that none of our (mental) re-presentations of "reality", whatever it may be: theory, vision, plan, concept, paradigm, hypothesis, expertise, conviction, ideal, politics, ... is totally in line with "reality" and even less with "Truth"!
Everyone tends to construct and communicate only "his" reality and some of the most eminent scientists do not escape this trap. It was the physicist Max Planck who considered that " a new idea never triumphs, but that opponents end up dying"
A particular limitation which constitutes in this context one of the major obstacles to the perception, understanding and mastery of complex situations that our contemporary societies have to manage is that all our intellectual training remains too much impregnated with the Cartesian paradigm which is, in essence, reductive, dissociative, logical, static, hierarchical in the face of an irreducible, global, networked, dynamic, dissipative world.
And if complementary approaches and methods and more integrative - not to say "systemic", this word still so often considered pejoratively by many so-called "rational" minds - have been developed and have proved their worth, they are still not - or so little - taught ... In his book "...", he says that the "systemic" approach to the problem of the "rational" mind is not only a question of the "rational" mind, but also of the "rational" mind. The Macroscope "Joël de Rosnay nevertheless clearly explains the necessary complementarity between the two approaches (figure 1).
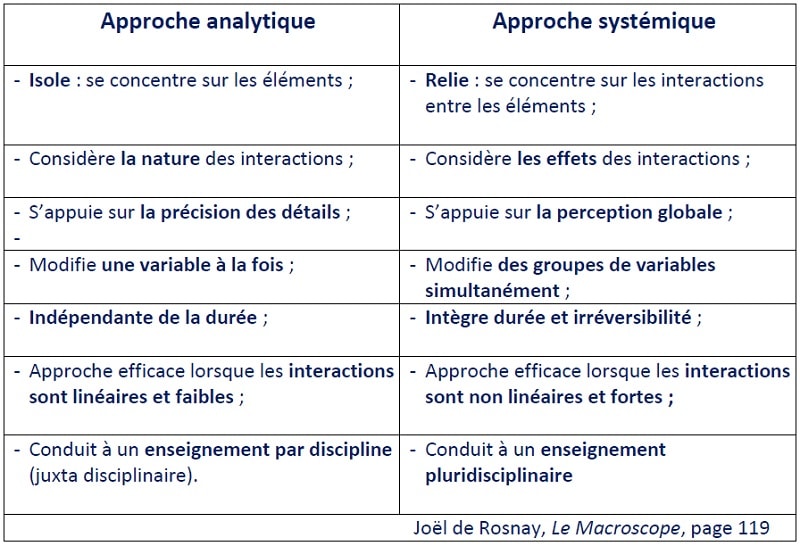
Often it is the lack of knowledge of the method for integrating the analytical and systemic vision of complex processes as a whole that makes it difficult to evaluate them and the issues involved. In many areas, this leads to debates of opinion that sometimes stray from the facts, making reasonable regulatory decisions difficult.
Why not enjoy unlimited reading of UP'? Subscribe from €1.90 per week.
The "cartoon case" of the glyphosate debate illustrates this lack of an integrative view of objective facts: for example, its classification by the International Agency for Research on Cancer (IARC-WHO) as " proven carcinogenic for animals and probable for humans ("Class 2A") is based, in its own words, on its intrinsic property of danger carcinogenic, a concept of danger that we will define later. On the other hand, classification by the European Food Safety Agency (EFSA) is based on the level of risk (the probability) of being exposed to this danger, which is a different and complementary notion to which we shall also return. Contrary to what is too often proclaimed, the classifications of these two agencies are therefore in no way contradictory!
Between facts and opinions, the evolution of civil society's expectations of regulatory decisions
In recent decades, there has been an evolution of civil society that has multiplied the interventions of its various stakeholders in the decision-making and regulatory management processes (Figure 2).
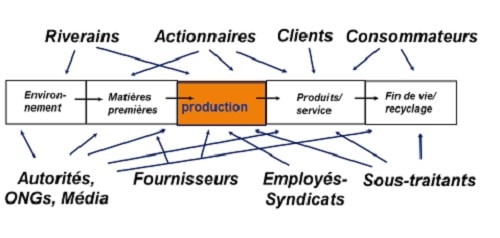
In such a context, it is often difficult to bridge the gap between the justified expectations of civil society with regard to information on the dangers and risks associated in particular with chemicals (manufacture, transport, uses, etc.), and the necessarily rational and factual responses to be provided by those who are "responsible" for them. These expectations are indeed not devoid of "emotional" dimensions to which these "technocratic" responses are sometimes considered abstract and frustrating, especially at a time when facts and "opinions" are, deliberately or not, mixed up and confused (Figure 3).

This complementarity of a dissociative and integrative approach is, however, an element essential in the investigative approach in general and toxicological, or even more, eco-toxicological, in particular: if toxicology is central to thes proposal for a Directive on the protection of a single species, the human species. and on and protection of the individual human being based on an extrapolation of the results of all tests and experiments on different species of animals, ecotoxicology, on the other hand, is focused on the protection of of all species in their structures, functions, populations and communities, ecosystems and all their interactions, based on a limited number of tests and experiments.
What is the overall strategy for assessing the hazards, risks and safety associated with chemical exposure?
What is proposed here is then a brief description of the integrative and realistic method that is applied today at European level for the assessment of hazards and risks related to the use of chemical or biological substances:
- identify intrinsically hazardous properties chemical substances for humans and/or the environment ;
- risk-based assessment associated with the forms of exposure to it/them;
- integrate "external" factors such as the risk/benefit balance sheet related to their usefulness, but also their societal acceptance (e.g. tobacco), cultural (ex: consumption of meat, alcohol, gluten, ...), politics (e.g. weapons), ethical or philosophical (e.g. animal protection) ;
- identify technological constraints s related to it (e.g. product substitution "(e.g. substitution of ozone- or climate-destroying refrigerants, pesticides);
- decide on a safety level societally "acceptable" (e.g. limit concentrations of contaminants) ;
- Case Management where the available information is (still) insufficient only to be able to make decisions on an objective basis and to be able to take precautions.
To find your way through this process, which allows us to identify on the one hand the properties inherently hazardous ( agent's "dangers" ) be it chemical but also physical or biological), and, on the other hand, to assess the risks associated to being exposed to it and to help determine the levels of risk which, under specific conditions of exposure, may or may not be considered acceptable or "tolerable", it is useful to distinguish four complementary but often confused poles (Figure 4).
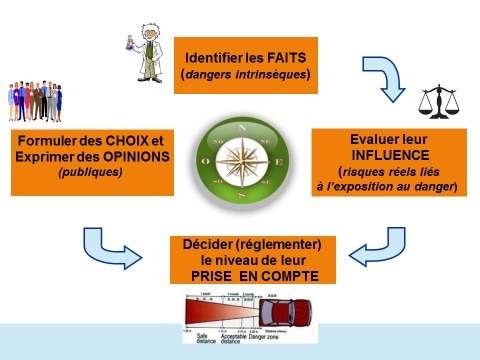
1st pole: Identifying the dangers
The concept of "hazard" describes undesirable properties intrinsically associated with the nature of an item: salt is corrosive, speed is dangerous, some radiation, dioxin or alcohol are toxic. This dangerous character depends on the intensity of the exposure which is the combination of the dose and duration or frequency of exposure to this adverse reaction. For most chemical, but also biological and physical (waves, radiation, noise, for example), (eco)-toxicological analyses and tests are used to determine the type of effect, and thus the hazard, and a "no-effect level". A toxic effect in this context is any "undesirable" effect.
According to the sensitivity There are, however, specific differences such as, for example, those of anti-infectious agents or pesticides in relation to their targets: bacteria, insects, ... ; or, within the human species, differences in sensitivity according to ethnicity, sex or individual profile (genetic, epigenetic, physiological and metabolic), age, belonging to vulnerable or sick populations.
Differences in the physiological processes of elimination and excretion of substances, whether natural or exogenous, can thus significantly influence the intrinsic toxicity of a (bio)chemical agent.
To fight against disinformation and to favour analyses that decipher the news, join the circle of UP' subscribers.
Within an organization specific enzymes are designed to promote the excretion of chemical substances, both those with physiological activity such as hormones and those absorbed by the body (e.g. alcohol), by modifying their structure to make them more soluble in waterand therefore in the urine. These enzymes are mainly active in the liver, but also in the kidneys, lungs and gastrointestinal tract.
It is through this metabolic process that the body eliminates exogenous substances that are poorly or not at all soluble in water and that can accumulate in cell membranes and fats. For example, by converting water-insoluble benzene into easily excreted phenol. Paradoxically, this process is nevertheless the one that sometimes generates the truly toxic and/or carcinogenic substances. Such as by forming DDE from DDT or the "epoxy" forms of benzene or that of benzopyrene, the polycyclic hydrocarbons present in cigarette smoke, which are the intermediate substances that are truly toxic and carcinogenic. It should be noted that the identification of the individual metabolic elimination profile is today also increasingly taken into account to optimise chemotherapy treatments, which are often highly toxic.
The intrinsic hazardous properties of a substance are therefore characterised on the basis of different types of experimental toxicological data obtained by a series of predefined tests. These databases are mandatory to obtain marketing authorisation as the European REACH Regulation and include acute and chronic toxicity (resulting from prolonged exposure of laboratory animals) by the oral and, where appropriate, inhalation routes of exposure: and define a "...". no-effect level (undesirable)"; they also include specific data on their carcinogenicity, mutagenicity and toxicity for reproduction. ( ("CMR"): where appropriate for substances already on the market or present in the environment, additional epidemiological (human population exposure analysis) and/or toxicological data.
Toxicology is therefore a multidisciplinary science by nature which involves, in order to gather these data, the multiple disciplines of both human and veterinary pharmacomedical sciences:
pharmacology, pharmacokinetics, (histo)pathology, haematology, ... ; analytical toxicology, " in vivo "and "in vitro "Experimental toxicology and clinical toxicology: acute and repeated dose toxicology: subacute, subchronic, chronic, multigenerational; clinical chemistry, cell toxicology, genetics (and "omics"); immunotoxicology, reproductive toxicology, oncology; toxicology of mechanisms of action and epidemiological toxicology; bio-statistics and mathematical modelling.
Toxicology then applies to the evaluation of all agents, whether physical (waves, radiation, ...), chemical or biological, including of course drugs and/or medical devices and industrial chemicals and plant protection products (insecticides, herbicides, fungicides, ...), additives and contaminants, all substances potentially or actually present in food products and packaging materials, consumer goods, industrial and household products, cosmetics and personal care products at the manufacturing, distribution, use or waste stage and, beyond this, genetically modified organisms,
To assess the potential toxicity of these agents to the environment and ecosystems, their potential toxicity will be identified in a laboratory and reproducible manner on representative model organisms of each trophic level. These basic tests are carried out on aquatic organisms, which are more easily standardised and reproducible, and have demonstrated their truly representative character :
- for plants: micro-algae (blue-green algae, diatoms);
- for crustaceans and molluscs: daphnia ;
- for mammals: fish : (rainbow trout, exotic aquarium fish);
- for microorganisms in charge of biodegradation: specific bacteria or wastewater treatment plant "sludge" samples.
For products deliberately intended for release into the environment (plant protection products especially) or presenting a particular concern, additional tests are carried out on other target species: birds, terrestrial mammals, bees, ...
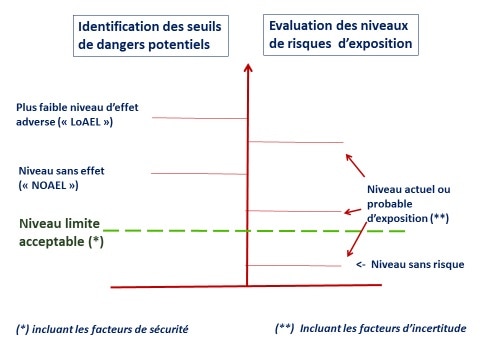
In order to define the acceptable limit value for exposure to a substance, natural or otherwise, with potentially dangerous or undesirable effects, the results of the abovementioned battery of tests shall be used to establish the threshold concentration at which these undesirable effects may occur.
A margin (or factor) of safety is always included, a margin that takes into account possible differences between species, between individuals and also differences between observations under experimental conditions and the diversity of real, human or environmental conditions. This factor is generally between 100 and 1000, depending in particular on the number and types of test studies carried out, the type of effect (irreversible or not), the degree of reliability of the knowledge acquired on the hazardous properties of the substance concerned (Figure 5 ).

In the case of the risk of collision on motorways, the safety factor is 2: 2 strokes of 25 metres or 50 metres. All things being equal, if the factor of 100 applied to chemical substances were applied, this makes it possible to appreciate what the safety distance between two vehicles would be: 2.5 km
Hazard assessment methods take broad consideration of potential effects that provide the highest level of concern, such as immunological effects, respiratory sensitization, carcinogenicity or reproductive toxicity. In order to address the danger of potential toxicity of mixtures of substances (the so-called "cocktail effect"), the rule of additivity of doses is generally applied, as many studies have shown that in most cases this reflects the reality of the situation, in spite of ongoing debates on the subject.
At present, the debate is particularly exacerbated by issues related to low-dose exposures to substances suspected of exerting so-called hormone disrupting effects: the so-called "endocrine disrupters" (EDs). However, the exact nature of the modes of action of these effects is still unclear, even when a link has been demonstrated between an adverse effect and exposure to a specific ED. The period of exposure is an important factor, such as in the womb or during the early stages of neonatal life. And sometimes these are exposure situations that are difficult to extrapolate to the whole population.
The current difficulty in defining hazard thresholds for substances producing this type of effect is largely due to the potential multiplicity of such effects and the lack of a reliable battery of (eco)toxicological tests that would allow a clear identification of these hazards. The same problem arises with regard to "nanomaterials". We will see below how to deal with these particular "uncertainties" about the very nature of a hazardous property.
2nd division: Risk assessment
It is then a question of establishing the relationship between the danger intrinsic nature of a substance and the risk potential to be exposed to it. This is a critical stage that is often misinterpreted because of the confusion, (sometimes deliberate or not) often existing between the two notions.
The risk is therefore related to the level of exposure to an agent with hazardous or undesirable properties; the degree of risk then depends on a combination of the frequency exhibition and the intensity of the exhibition:
Risk = Danger x Exposure
Risk is therefore defined as the probability of exposure to a hazard. The actual (measured or calculated) or expected levels of exposure are compared to the hazard thresholds (Figure 6). This is done by incorporating the degree of uncertainty included in the safety factors mentioned above but also the characteristics of the potential exposure level of specific populations. ( who??) » : professionals, general public, ecosystem) and also integrates a "geographical" dimension ("where??": factory, housing, public places, ...).
Unlike a danger which is intrinsic to a substance and cannot be modified, the risk of exposure can often be controlled by source prevention or protective measures, by analyzing the "how?" and "why?" of this exhibition.
For chemicals with different modes of action (acting independently), there is no strong evidence that exposure to a mixture of these substances produces "multiplier" effects and constitutes a health or environmental problem if, overall, the levels of exposure to all of these chemical compounds are below their no-effect levels. While many toxicologists have dreamed of making a career out of establishing such multiplicative effects[1] ("cocktail effects"), in the many cases studied more specifically, it has indeed been established by several more recent reports that the addition of doses (or concentrations) overestimates even slightly the actual toxicity of their mixture. This suggests that, on the whole, the use of the concept of additivity of doses or concentrations for the assessment of health risks related to chemicals, even via as yet unknown mechanisms of toxicity, is overall sufficiently protective.[2].
|
The case of substances suspected of exerting hormone-disrupting effects. At the low levels of exposure encountered, there is still only limited evidence today that some of these substances have a potential hormone disrupting effect (danger) are responsible for risks for the health problems encountered in men: reduced sperm quality and fertility, increased frequency of certain types of cancer, increase in the number of spontaneous abortions, congenital anomalies of the sexual organs, ... Experimental studies indicate that such endocrine system disorders occur at exposure levels that appear to be higher than current or established acceptable exposure thresholds. However, these effects justify the concern that simultaneous exposure to different substances with hormone-disrupting properties may produce undesirable effects, sometimes even at the low levels of exposure encountered for each substance individually. In this context, the European Union has drawn up a list of 300 priority substances with endocrine disrupting potential, which shows that this issue is taken very seriously and the debate remains particularly exacerbated by questions relating to the potential effects of "endocrine disrupters" at low doses. For effects on ecological systems (the "environment") in particular, however, one report indicates that exposure to mixtures of dissimilar substances acting at low, but potentially relevant, concentrations should be considered a potential concern, even if each of the substances is present individually at "..." (see below).predicted no-effect concentrations” (Predicted No Effect Concentrations(SEE "PNEC"). There is therefore a continuing need to improve experimental methodologies and knowledge, and to develop more integrative or systemic approaches to ecological risk assessment under realistic conditions of the multiplicity of interactions associated with environmental exposure to mixtures of chemicals. |
3rd division: safety and risk management
Since the risk of exposure to potentially hazardous agents can be reduced and therefore often controlled, based on anticipated, assessed or measured levels of exposure, such risk reduction measures can be identified, proposed and implemented. : modification of the production process, dilution of products, restrictions on use, user training, isolation and control of emission sources, personal protective equipment in the workplace: helmets, clothing, masks, ventilation, hygiene, etc.
- In industry, risk control also includes the design and conduct of strategies and complementary toxicological studies and precise documentation on the hazards and risks associated with product safety: files, safety data sheets and material safety data sheets, classification and labelling, standard exposure scenarios in certain applications, etc.
- In medicine, it will be the evaluation and monitoring of individual exposures, including for side effects of drugs, medical monitoring and emergency medicine, management of "poisons", forensic medicine, ...
- In everyday life, safety measures range from product labelling to restrictions on use in non-professional applications, from safety caps on bottles of dangerous products to protective equipment, ...but also for other types of risks from water disinfection to bumpers or seat belts, fire extinguishers in homes, retention basins under oil tanks, ...
It is these innumerable measures that in most cases make it possible to benefit from the often significant advantages of many products and their applications by limiting their undesirable effects to an "acceptable" level.
A level of safety that is considered acceptable must also incorporate trade-offs between the advantages and disadvantages of using certain substances, such as the technological ability to control risks or the socio-economic constraints of foregoing their "benefits" (ex : drugs, anti-parasite products, preservatives, but also mobile phones, fossil fuels and even alcohol or tobacco ...). This therefore implies also taking into account a series of social and societal, even cultural or ethical references: political, philosophical or even emotional choices ...
Thus, the risks considered "acceptable" will not be the same for trained and protected professionals and for simple "residents" of a factory or field; for medicines and food; for drinking water and for alcohol or tobacco; for automobile and aeronautical risks; on the normal road or in a rally; in countries more or less exposed to other risks (infectious diseases, food safety, for example), in situations of war or peace, ...
While in some cases, risk control measures seem insufficient to meet the levels of risk defined as acceptable, other options, such as substitution of the substance involved, should be considered. This has been the case in recent decades for a large number of substances such as solvents, organochlorine pesticides, the much-disputed glyphosate, food additives or the notorious CFCs (chlorofluorocarbons) ozone depleting substances and, in particular, for substances combining the three properties of Persistence in the environment, Bioaccumulation and Toxicity : PBT" substances »).
Safety is therefore not defined by absolute criteria and the decision defining a risk as "acceptable" is not (only) based on scientific or medical considerations: it is a safety limit that society chooses at a given time to consider acceptable: it is a societal and therefore "political" responsibility.
4th pole: perceiving risks and expressing one's opinion
The risk perception is not necessarily related to its objective significance. The level of safety adopted will therefore be more or less accepted by citizens depending on the perception of risk and its acceptance.
Accepting a risk does indeed imply a subjective and emotional dimension (figure 7); some people sometimes play on it or even abuse it demagogically ("disinformation").
However, an opinion or "conviction", once formed, is very difficult to change.. And the problem is that, too often, opinions are formed independently and prior to the knowledge of the facts that are supposed to feed them....
- "accepted" risk: smoking, skiing, drinking, ... ;
- "tolerated" risk: road, medication, ... ;
-risk "undergone": food, water, plant, pesticides, nuclear . . ;
-natural" risk: familiar, accidental, memorable, spectacular.
Figure 7: The relative perception of "risks" by individuals
However, an advantage or "benefit" is the expected result of any activity that presents certain risks: to eat, you had to hunt or work; to heat, you had to cut wood; to sell, you had to manufacture; to preserve your health (and that of others), you had to get vaccinated; for your well-being, you had to practice a sport, sometimes "dangerous".
It is therefore upstream of the opinions that the "scientific" facts explaining both the dangers and risks, but also the expected benefits, must be submitted to the stakeholders. These facts must, of course, be synthesized and made available in a language accessible to non-specialists:
- faithful and strictly factual, including within the limits of their interpretation;
- simplified in their expression (no jargon!) validated by peers.
How, then, can we better balance and (make) take into account both the facts, their context and concrete meaning, and help to form opinions on this basis for important regulatory decisions? This is a key issue if policy makers are to propose and support regulatory measures that are reasonable, effective and well accepted in their application and contribute to objective and factual information about hazards and risks.
Fith regard to uncertainty, the Precautionary Principle ...
Some substances, as we have seen, still sometimes resist the capacity to analyse their intrinsic dangers, risks and consequences: "mad cow" or "dioxin chickens" at one time and today, in particular the glyphosatethe fipronil used in chicken farming, the sulfoxaflor and neonicotinoids toxic to pollinating insects, the chlorpyrifos used to control aphids, caterpillars and seed flies or endocrine disruptors, certain nanomaterials but also certain electromagnetic waves, certain GMOs or in particular future applications of "synthetic biology".[3].
The objective is then to "manage uncertainty" with caution and to be able to decide and act in a regulatory manner when the "experts" cannot (yet) give a formal opinion. The problem in practice is that the measures taken must be proportionate to the risk envisaged.
Visit Precautionary Principle as defined by the European Union[4] and recognised by its mention in the Maastricht Treaty states that : "Where there is sufficient evidence to suggest that an activity is seriously suspected of causing irreversible damage to the environment or health, action must be taken, even if the ultimate scientific evidence or causal link is not yet formally established with certainty. ”
In some Member States, such as Belgium and the Netherlands, the courts recognise the precautionary principle provided that it has been enshrined in a specific law, and in France it has been incorporated into the Constitution.[5]. It is then possible to apply it and ban, on a transitional or prolonged basis, the use of a product meeting its criteria.
...and that of Proportion!
But to do so, and as explicitly recommended by the "...". courses of action "In order to be able to apply at the same time another fundamental principle of our "law" inscribed in the texts: the Principle of Proportion!

Considering the substitution of an agent is a typical example of the need to apply this indispensable balance to be respected, between Precaution and Proportion in the more or less significant probability of undesirable effects (risks) related to properties of a product to manifest themselves. When the question arises of the substitution of a substance with "undesirable" properties, it is however legitimate, even essential, to also consider whether its properties "may be affected by the use of a substance". desirable "do not justify uses" indispensable "It's just as important for our health or the environment or even the lifestyles we aspire to.
It is therefore a question of ensuring that possible substitutes guarantee the maintenance of these beneficial effects in applications considered essential to our well-being ... For example, fire, oxygen, bottled gas are certainly dangerous in themselves, as is the risk of over-consumption of sugar or salt, or the use of antiseptics and antiparasitic drugs such as DDT, which is still irreplaceable in the face of malaria, which still kills...[6]and a whole range of indispensable products. Faced with this, tobacco and alcohol and, increasingly, cannabis for example, whose undesirable effects are no longer to be proven, remain ignored in these substitution procedures ...
Regulatory management of the hazards and risks of chemicals at the European level
To manage the hazards, risks and safety of chemicals at the European level, the REACH Regulation adopted in 2006[7] improved and harmonised the provisions relating to knowledge of the properties of chemical substances manufactured or placed on the market in the European Union, those intended to ensure control of the risks associated with their use and, if necessary, to reduce or prohibit their use. For all chemical substances, any company that manufactures, imports or uses them must file a registration dossier with the European Chemical Safety Agency (ECHA). Lighter registration procedures are applied for certain substances at the research stage or only used as intermediates in the manufacturing process.
An annexed list lists substances of "concern" or those that present a risk considered "uncontrolled" and their uses are then subject to a specific and transitional authorisation procedure aimed at possibly limiting their manufacture, use or placing on the market, or even prohibiting their use.
Conclusion
Well-established facts and ongoing dialogue between stakeholders are essential to arrive at relevant regulatory decisions on the use of chemicals. Reasonable decision making requires a constant dialogue between all stakeholders, which contributes to the construction of opinions that integrate a contextualization of the different elements.
Indeed, democracy is based on the transparency of the choices in which citizens now want to be seen as partners and not just as mere "governed" or "cumbersome neighbours". Indeed, they expect products to be surplus valueservices that meet their needs, expectations and desires. Some of these "expectations" (food, health, transport, energy, housing, tobacco, speed, but also cost price, employment, etc..) will only be met with certain disadvantages (risks); how willing are we to forego these benefits because they are sometimes unsustainable?
The challenge is therefore to provide citizens with the means to understand and actively participate in decision-making processes. But, in turn, to power To assume this responsibility, the citizen must know theexercise and assume its duty of lucidity in the face of the facts and opinions submitted to it. In an environment increasingly exposed to the sometimes most esoteric opinions that some networks, and in particular some so-called social ones, try to inflict on them (" fake news "and others ...), so avoid relying passively on ready-made, simplistic and sometimes demagogic positions. And do not forget that, whatever the influence attributed to the "lobbies" of all origins, it is the "elected representatives" of the citizens who bear the real responsibility for decisions by pushing for ultimately on the voting buttons in the assemblies...
It is the construction of this permanent pedagogical dialogue that will enable each citizen to be ready to: avoid purely emotional "excess"; distinguish between danger, risk and security; weigh risks and benefits; understand that accepting a risk implies choosing it; put into perspective precaution and proportion; have more confidence in factual arguments than in diverse opinions on the day when debates or crises arise.
Jacques de GerlacheDr. Sc Pharm, (eco)toxicologist
You can find a brief presentation of the notions of Danger-Risk-Security and Precaution in an animated video: https://www.youtube.com/watch?v=wRmfvFYDNr8
[1] There were even major scientific frauds in this field and the journal "Science" was officially asked to consider that a famous article it had published in the late 1990s on the supposed combined effects of organochlorine substances had not been!
[2] GreenFacts' proposed 'Highlights' of an EFSA report on the subject will be online on 15 September ...
[3] Highlights" reports for most of these substances are or will be posted on the site. www.greenfacts.org/fr/
[4] Communication from the Commission on the precautionary principle, European Commission, COM(2000)1. http://ec.europa.eu/dorie/fileDownload.do;jsessionid=pTLtRm1Pz8hXQQpYClb609jmM3VTQqJvm1WZ9TT701CWBXyWQcyY!600366969?docId=134617&cardId=134617
[5] https://www.encyclopedie-environnement.org/zoom/principe-precaution/ http://www.europarl.europa.eu/RegData/etudes/IDAN/2015/573876/EPRS_IDA%282015%29573876_FR.pdf
[6] In 2015, according to WHO, there were still about 212 million cases and some 429,000 deaths due to this disease. http://www.who.int/features/factfiles/malaria/fr/
[7] REACH: Registration, Evaluation, Authorization and restriction of CHemicals- Registration, Evaluation, Authorisation and Restriction of Chemicals ? In 2006, the European Union Regulation updated all European legislation (more than 40 directives) on the manufacture and use of chemical substances through a single integrated system for the registration, evaluation and authorisation of chemical substances. https://echa.europa.eu/fr/regulations/reach/understanding-reach












