Aoestern medicine, based on evidence, is a rational art derived from the knowledge of the causes and mechanisms of diseases. France stands out for its technologically advanced health system, accessible to all in a principle of solidarity and equity. However, it is confronted with a constrained economic model and difficulties due to the compartmentalization of medical specialties and insufficient links between the hospital, town medicine and social actors. Moreover, it is only recently that patient care has been trying to move away from reference systems based on average data, which erase the specific characteristics of each patient, towards precision medicine, which is the new paradigm of care.
Our current medicine probably also gives insufficient room to prevention, not because of political will, but because of the difficulty of convincing citizens. It is to be hoped that the medicine of the future, in France and elsewhere, can become more predictive, preventive, participatory and precise. The overhaul of the organisation of the healthcare system will help to decompartmentalise it and to better define the place of real and virtual players around the patient in a vision of health democracy.
Exploring the genome, metagenomes and exposome for precision, predictive and preventive medicine
Genome, metagenomes and exposome shape who we are and largely condition the prevalence and particularities of each person's diseases.
The crucial place of the genome and its epigenomes in individual development and self-definition has long been experimentally validated. However, it is only in recent decades that these data have invaded health research and the practice of medicine. From the key areas of bone marrow and organ transplantation, rare single-factor genetic diseases and cancers, the interest of researchers has shifted to other common multifactorial diseases.
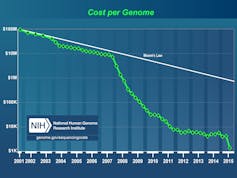
The cost of sequencing a genome has dropped drastically in 15 years. NIH/DR
The weight of genes in prevalence and clinical forms is beginning to be established against the backdrop of the complexity brought about by the genetic variations between populations. This field of research has been overturned by technological developments and the lower sequencing costs that bring the clinical use of genomic approaches within the reach of the physician and facilitate the interpretation of those approaches - transcriptomics, proteomics, metabolomics.
Avoiding the diagnostic wandering that afflicts families suffering from genetic diseases, adapting cancer treatment on the basis of the individual's genome and tumour, better understanding and perhaps predicting and preventing common multifactorial diseases, this is the roadmap that France has set itself in the Plan France Médecine Génomique 2025 (PFMG).
Until recently, researchers and doctors have reappropriated the notion that man or other living elements are symbiotic beings. It would be artificial to consider man independently of the micro-organisms that populate the intestine, skin and mucous membranes. These micro-organisms exchange signals with our organism and play decisive roles in many diseases such as obesity or certain neurological or psychiatric illnesses. These germs can also be critical to the effectiveness of therapies, such as immunotherapy for cancer. Assessing the diversity of our microbiotes by "-omics" approaches could usefully complement the PFMG tools in the future.
If genomics defines the field of possibilities for each individual from conception, what we are at all stages of life is largely determined by our exposure to the outside world through our lifestyle and its social determinants, our work environment and more generally by the environment in general, which has been largely shaped by man since the industrial period. The body is shaped by food (which can by extension be considered as exposure). It is altered by exposure to toxic substances or by encountering infectious agents or their vectors. In this context, it is clear that human health cannot be isolated from a more global health that includes all living things ("One Health").
Collecting and integrating these data with clinical elements and quantitative data from in vivo clinical imaging is a major challenge for the precision medicine of the future. This can only be done by developing a specialized industrial sector of medical devices to interrogate the genome, theepigenome and the exposome, a sector for which the PFMG's Centre of Reference, Innovation, Expertise and Transfer (CREFIX) should be a first brick and a major orchestrator.
Putting the patient, the doctor and their avatars in a clinical situation for participatory, transparent and equitable digital medicine
Many organizational schemes of individual medicine are possible, even the disappearance of health professionals replaced by algorithms and diagnostic and care devices provided by industrial companies from a global catalogue. Less ambitious but guaranteeing equity, the scenario I suggest retains a relationship between patient and doctor, sentient beings, while incorporating technological companions that optimize the common approach.
In this model, the patient is associated with a digital representation or digital twin and a biological companion that groups together all the personalized resources extracted or constructed from his or her body. The doctor, for his part, is supported by the digital representation of his knowledge and decision rules (digital doctor). He is also associated with sensors and diagnostic tools that build the digital patient by converting biological, imaging or clinical data into digital files.
In order to intervene on the patient, the doctor has at his disposal classic approaches (drugs, surgical acts, physical agents, rehabilitation, psychotherapy), enriched with emerging technologies. Digital doctor and digital patient in interaction with the knowledge space, optimize the decision support shared by the patient and the doctor who, freed from technical details, should have the necessary time to find a common language in a principle of mediation, transparency and equity.
Building, with the industrial fabric, the three technological pillars of integrated medicine, a source of economic development
The medicine of the future will be very strongly impacted by three technological pillars, major sources of research and economic development.
The first pillar is multidisciplinary approaches to the development of medical devices for diagnostic, therapeutic or mixed use. They will benefit from chemistry, synthetic biology, various physics including electronics and materials science, robotics, nanotechnologies and information sciences.
The second pillar is that of the biological companion, which brings together all the personalized biotechnological developments built from elements of the patient's body, such as the development of organs on chips or the genetic modification of cells for precision medicine.
The last pillar, finally, is digital, integrating the medicine of the future and building a self-learning health system.
Hybrid nano-objects, live-electronics and robotics interfaces
Nanotechnology already plays an important role in medicine. In the field of diagnostics, they are crucial for increasing the sensitivity of in-vivo imaging, whose ultimate goal (especially for magnetic resonance imaging) is visualization at the cellular level. In therapeutics, nano-objects are used for the delivery of active ingredients or to increase the effectiveness of radiotherapy whose modalities evolve with technological and radiobiological advances.
The possibility of coupling synthetic biology and electronic devices within the same object could open the way to multifunctional communicating objects, both sensors of biological and physical data and therapeutic tools delivering active principles in a reasoned way. In this context, the development of biocompatible and, where appropriate, biodegradable electronics will be a key step.
The ageing of the population is increasing the number of patients with degenerative diseases for which therapies remain ineffective. These diseases lead to major handicaps. In addition to providing substitute organs, such as artificial retina, or exoskeletons, medicine will have to support disabled people to improve their well-being in their usual environment. The design of robots to help them in their daily lives will be crucial in this context.
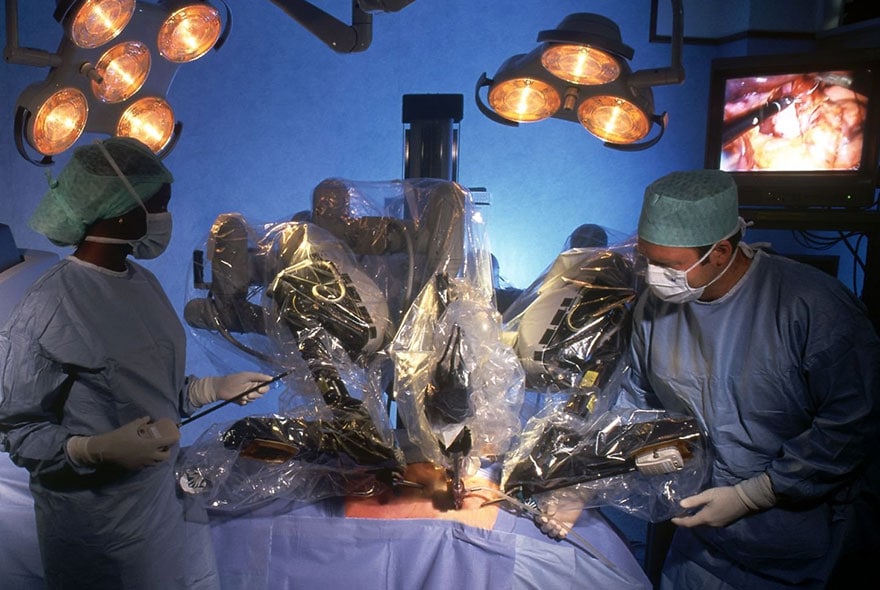
In addition to technological developments, the elaboration of these robots will require important developments of artificial intelligence algorithms participating in the creation of a personalized, intelligent and connected living space. Increasingly, remotely controllable robots will accompany surgeons in a less and less invasive practice, benefiting from an increased vision of the operating field and a personalized preoperative simulation based on the patient's imaging data.
Biological companion of the patient and reconstruction of tissues and organs
Significant advances in stem cell research are making it possible to obtain in vitro virtually every cell type that makes up the body. Advances in biotechnology, particularly in 3D bio-imprinting, are paving the way for the reconstruction of tissues and organs that retain their biological functions. They also make it possible to reconstruct malignant tumours in the image of the patient's own.
At the individual level, these biological companions can be used for the optimization of personalized treatments by studying ex vivo the effect of therapeutics, the data obtained enriching the patient's avatar. Reconstituted organs can also be re-implanted in the donor patient without immune rejection, reducing the need for cadaveric organs.
The constitution of collections of a large number of biological companions annotated by the genome sequence of the individual, will make it possible to simulate therapeutic trials. ex vivo and will reduce the risk of toxicity and inefficiency.
Digital medicine, the integrator of the medicine of the future
The ability to convert virtually all medical data (e.g. clinical, genomic, imaging) into digital data that can be understood by a computer (the digital patient) opens the way to a new medicine based on these data.
In this context, the doctor-patient relationship is a confrontation between the real world and a virtual world where three partners interact: scientific knowledge and data, digital representation of the patient (digital patient or digital twin) and artificial intelligence algorithms (or digital doctor). The latter interpret the digital patient and provide medical decision support by proposing precise diagnostic and therapeutic recommendations. The doctor becomes a critical mediator of information and remains the guarantor of the human character of this new medicine.
The inclusion of digital patient cohorts in a data collector and analyzer (such as the PFMG DAC) will allow the application of algorithms based on artificial intelligence, data science and simulation of biological processes that will guide the physician and patient in defining optimal management.
Everyone's data will be at everyone's service, contributing, through collective expertise, to personalized medicine and less dependent on the experience of a single doctor or medical team. In addition to disease management, this digital medicine will also apply to prediction and prevention, in particular via connected objects creating a health Internet.
It is likely that the question of sequencing the genome of all individuals will one day arise, subject to ethical debate and leading to the evolution of laws and regulations. Genome variability defines populations and is a heritage resource. Its analysis can make it possible to anticipate at the individual and collective level the ability to react to the surrounding environment. It is therefore data that must be stored in a highly secure location offering all the guarantees of against fraudulent or malicious use.
Imagining the innovations of the three pillars of the medicine of the future and bringing them to the patient requires effective interaction between healthcare professionals, academic and industrial researchers and companies that can bring them to market. Organisational innovations are needed. Proposals to this end have been made in the report. "Medicine of the Future".
François SigauxPU-PH, University Paris Diderot and APHP, Scientific Director, Commissariat à l'énergie atomique et aux énergies alternatives (CEA) - University of Paris-Saclay
This article is republished from The ConversationUP' Magazine's editorial partner.
Anything to add? Say it as a comment.