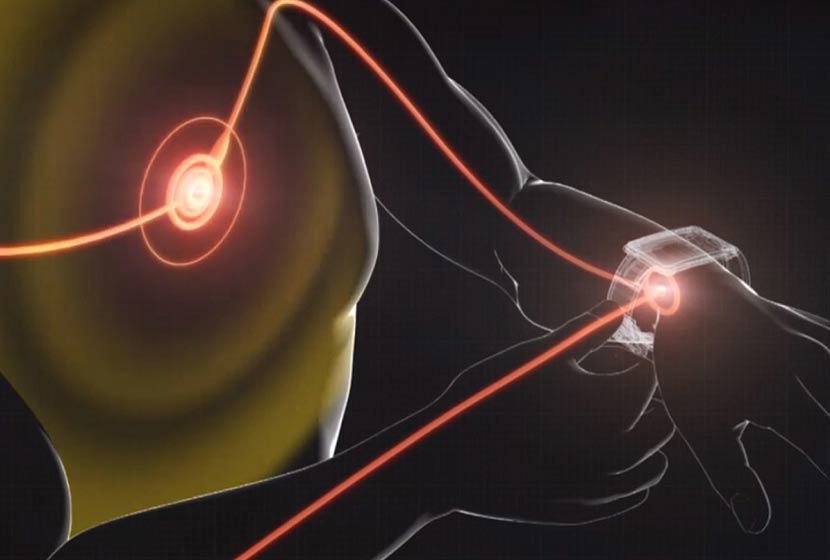
Bionic Eye: Canada's Ministry of Health approves Argus II, Second Sight's epiretinal prosthesis system, for the treatment of peripheral retinal degeneration.
Argus II is the first bionic eye authorized in Canada for the treatment of blindness due to peripheral retinal degeneration.
Already authorized for sale on the European market since 2011, the Argus II retinal prosthesis marketed by the Californian company Second Sight has just obtained authorization from the Canadian Ministry of Health (Health Canada) for the use of the Argus II prosthesis for the treatment of patients suffering from severe to major peripheral retinal degeneration.
Implantable visual prostheses to restore part of the visual function of blind patients.
The Argus II prosthesis is the only epiretinal prosthesis authorized by the Canadian Ministry of Health and the American FDA (Food & Drug Administration). The Argus II is also the first epiretinal prosthesis authorized in the world. The system induces visual perception in blind people via electrical pulses that stimulate the last living cells of the retina, creating a perception of light patterns in the brain. The visual abilities offered by the Argus II implant can change the lives of those who have no or no longer have visual function. Argus II can have a positive impact on the ability of blind people to carry out daily activities, such as recognizing shapes and large objects, locating people, locating the location of doorways, and being able to follow lines or edges. The Argus II therefore enables its users to be more independent in their daily lives.
"This is one of the most exciting medical discoveries I've witnessed in my career."says Dr. Robert Devenyi, Fellow of the American College of Surgeons, Chief of Ophthalmology at the Donald K. Johnson Eye Centre and Director of Retina Services at the University Health Network. "Argus II gives so much hope to patients with degenerative eye diseases. I am delighted to be part of the first Canadian team that can bring this technology to patients. » Dr. Devenyi implanted the Argus II on the first Canadian patient on June 5, 2014, at Toronto Western Hospital (Toronto) as part of a clinical trial.
"This regulatory agreement and the first Canadian implementations of Argus II are major milestones, not only for Second Sight, but also for Canadian patients with peripheral retinal degeneration."added Dr. Robert Greenberg, PhD, President and CEO of Second Sight. "We have more than 20 years of research and development and strong intellectual property. Following approval in the U.S., approval in Canada allows us to integrate other provinces, in addition to Ontario, into our international network of Centres of Excellence that offer the Argus II epiretinal implant. » Second Sight plans to include in this network other centres in large agglomerations and will apply for reimbursement of the prosthesis in the provinces to which these agglomerations are attached.
Already many patients in the world
 Second Sight's Argus II system received CE marking in Europe in 2011 and US FDA approval in 2013. It is the first epiretinal prosthesis to obtain these approvals worldwide. The Argus II has already been implanted in more than 90 patients. Many of these patients have been living with the prosthesis for more than seven years, demonstrating the long-term reliability of the device. The company has published 20 articles in peer-reviewed journals, including the most recent in BMC Ophthalmology: "The Argus II has been used in more than 90 patients worldwide, and many of these patients have been living with the prosthesis for more than seven years, demonstrating the long-term reliability of the device.The Argus II prosthesis facilitates reaching and grasping tasks: a case series." by Kotecha et al in BMC Ophthalmology 2014, 14:71.
Second Sight's Argus II system received CE marking in Europe in 2011 and US FDA approval in 2013. It is the first epiretinal prosthesis to obtain these approvals worldwide. The Argus II has already been implanted in more than 90 patients. Many of these patients have been living with the prosthesis for more than seven years, demonstrating the long-term reliability of the device. The company has published 20 articles in peer-reviewed journals, including the most recent in BMC Ophthalmology: "The Argus II has been used in more than 90 patients worldwide, and many of these patients have been living with the prosthesis for more than seven years, demonstrating the long-term reliability of the device.The Argus II prosthesis facilitates reaching and grasping tasks: a case series." by Kotecha et al in BMC Ophthalmology 2014, 14:71.
About peripheral retinal degeneration
Peripheral retinal degeneration is the deterioration of the outer layer of the retina. It results from the progressive death of cells located in this region of the retina. Pigmentary Retinitis (PR) is an important type of peripheral retinal degeneration. RP is a rare hereditary disease that causes a progressive degeneration of the photosensitive cells of the retina, leading to a significant deterioration of vision and may eventually lead to blindness. It is estimated that approximately 13,000 people in Canada and 1.2 million people worldwide have PR.
About the Argus II epiretinal prosthesis system
 Second Sight's Argus II system produces electrical stimulation to bypass dead cells and stimulate the remaining living retinal cells, which induces visual perception in people with severe to major peripheral retinal degeneration. Argus II converts images captured by a miniature camera mounted on the patient's eyeglasses into a series of small electrical pulses that are transmitted wirelessly to a series of electrodes implanted on the surface of the retina. These pulses are designed to stimulate the last living cells in the retina, resulting in the perception of light patterns in the brain. The patient then learns to interpret these visual patterns and regains some visual function. The Argus II is the first artificial retina to be approved in Europe (CE marking). It is also the first and only epiretinal prosthesis authorized in the United States and now in Canada.
Second Sight's Argus II system produces electrical stimulation to bypass dead cells and stimulate the remaining living retinal cells, which induces visual perception in people with severe to major peripheral retinal degeneration. Argus II converts images captured by a miniature camera mounted on the patient's eyeglasses into a series of small electrical pulses that are transmitted wirelessly to a series of electrodes implanted on the surface of the retina. These pulses are designed to stimulate the last living cells in the retina, resulting in the perception of light patterns in the brain. The patient then learns to interpret these visual patterns and regains some visual function. The Argus II is the first artificial retina to be approved in Europe (CE marking). It is also the first and only epiretinal prosthesis authorized in the United States and now in Canada.
About Second Sight
Second Sight Medical Products, Inc. was founded in 1998 with the goal of creating a retinal prosthesis capable of restoring sight to blind patients with peripheral retinal degeneration such as retinitis pigmentosa. Second Sight's mission is to develop, manufacture and market implantable visual prostheses to enable blind people to become more independent. Second Sight is also developing Orion, a cortical prosthesis to restore the sight of blind patients whose blindness is due to pathologies that can neither be avoided nor treated. Approximately 70,000 people are potentially eligible for the future Orion cortical prosthesis. Second Sight is headquartered in Sylmar, California and has European headquarters in Lausanne, Switzerland..