RACE FOR TREATMENTS :::: READ ALSO : The BCG really? / This vaccine could be a big hit. / Blood protection of healed patients / A marine oxygenating worm / WHO and Europe Mega-studies
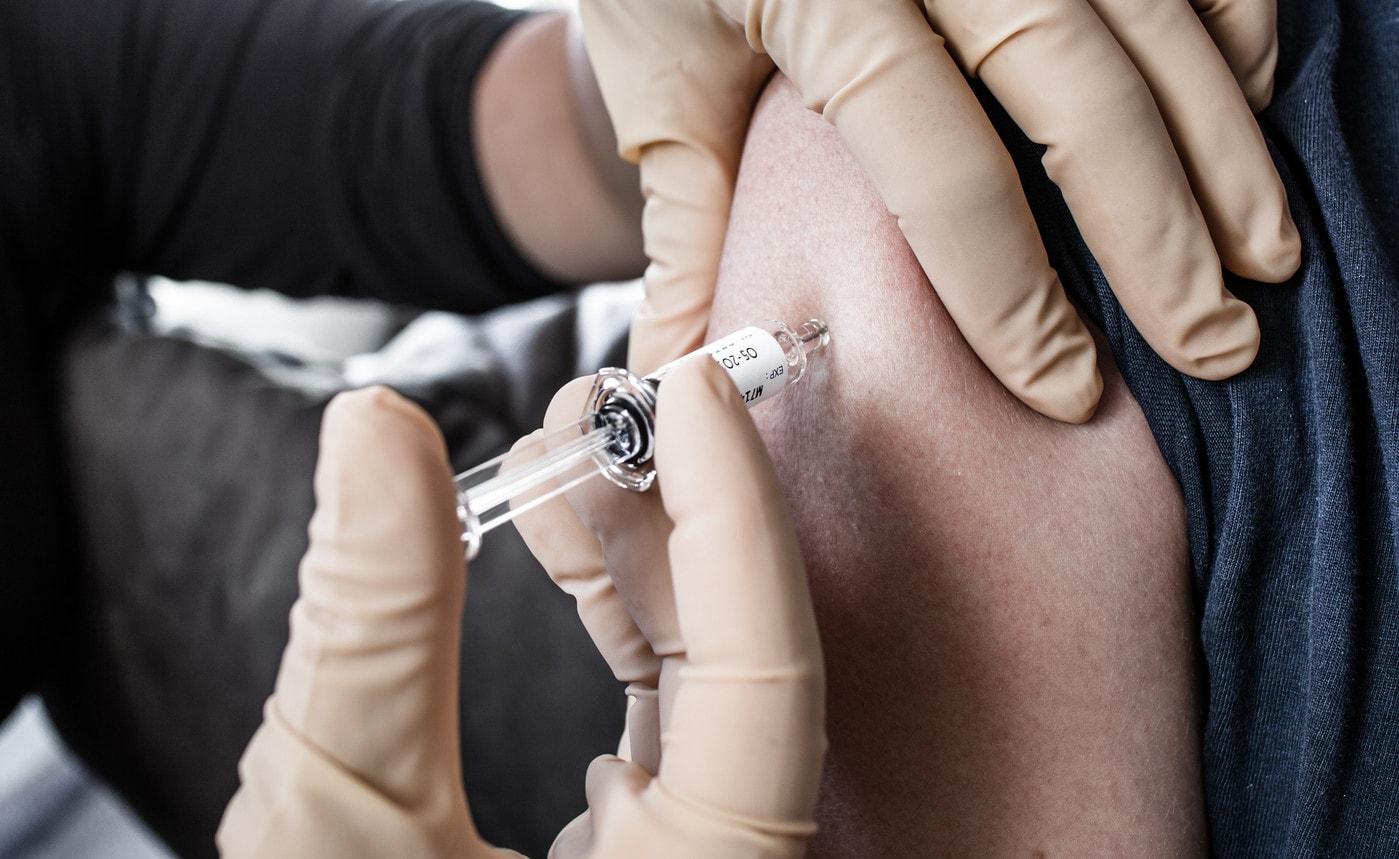
The United States has taken the option of having hundreds of millions of doses of vaccine manufactured, even before clinical trials have begun. They want to be ready for the market before others are. A risk in the form of a poker strike.
According to the agency ReutersThe US government has entered into agreements with Johnson & Johnson (J&J) and Moderna, and said it is in talks with at least two other pharmaceutical companies to prepare them to produce massive quantities of coronavirus vaccines before they are tested and safe and effective vaccines are available.
There is currently no treatment or vaccine specifically approved for COVID-19, the respiratory disease caused by the coronavirus, which has killed, at the time of writing, more than 68,000 people and infected more than 1,240,000 people worldwide in just a few months.
However, no vaccine should be ready for use before at least 2021, as it has yet to be widely tested in humans before being administered to hundreds of millions, if not billions of people to prevent infection.
On March 31, J&J announced a $1 billion agreement with the U.S. government to create sufficient production capacity to manufacture more than one billion doses of a vaccine. This agreement was reached at a time when the first tests on humans are not expected to take place, at best, until September 2020. Thereafter, experts estimate that it could take 12 to 18 months before a safe and effective vaccine receives regulatory approval.
In general, decisions to proceed with a vaccine are made after safety and preliminary efficacy trials have been completed. But J&J's chief scientific officer, Dr. Paul Stoffels, told Reuters that his company needs to start increasing production capacity now, even before there is a signal that its experimental vaccine is working.
" It's the only option for us to get it in time. "he says.
J&J has a manufacturing facility in the Netherlands that can produce up to 300 million doses of vaccine, Stoffels said, but "that's not enough. will be absolutely not enough for the world ». As a result, the company is also looking for manufacturing facilities in other parts of Europe and Asia capable of producing the type of vaccine it is working on.
According to Stoffels, J&J's vaccine will be based on the same technology used to make its Ebola vaccine, which has been widely used in humans. The company believes it will be safe. It's a gamble in the form of a poker shot.
Source: Reuters